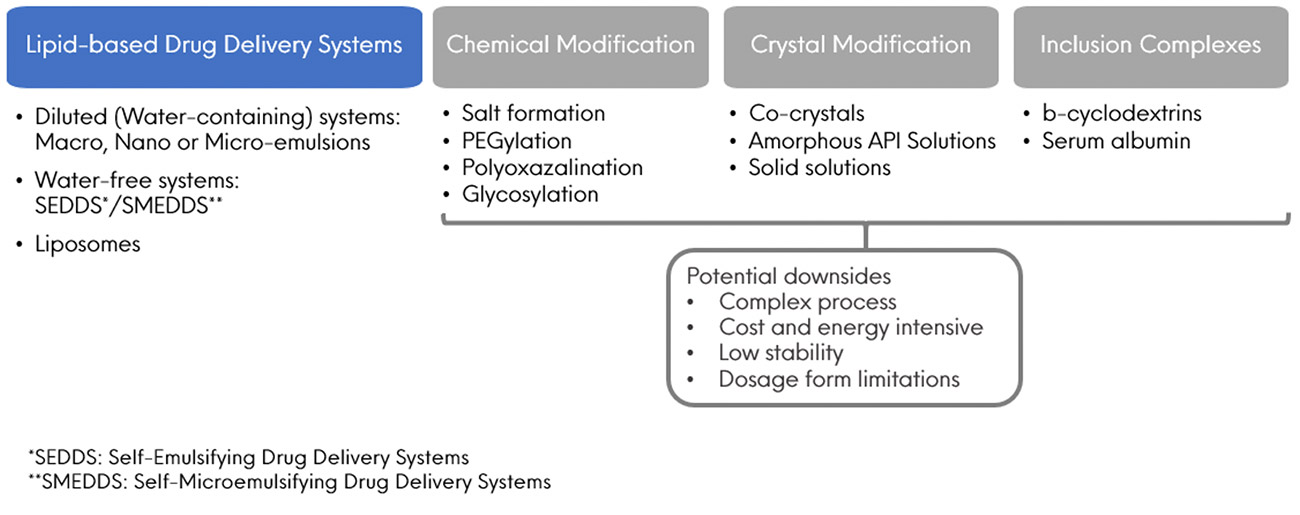
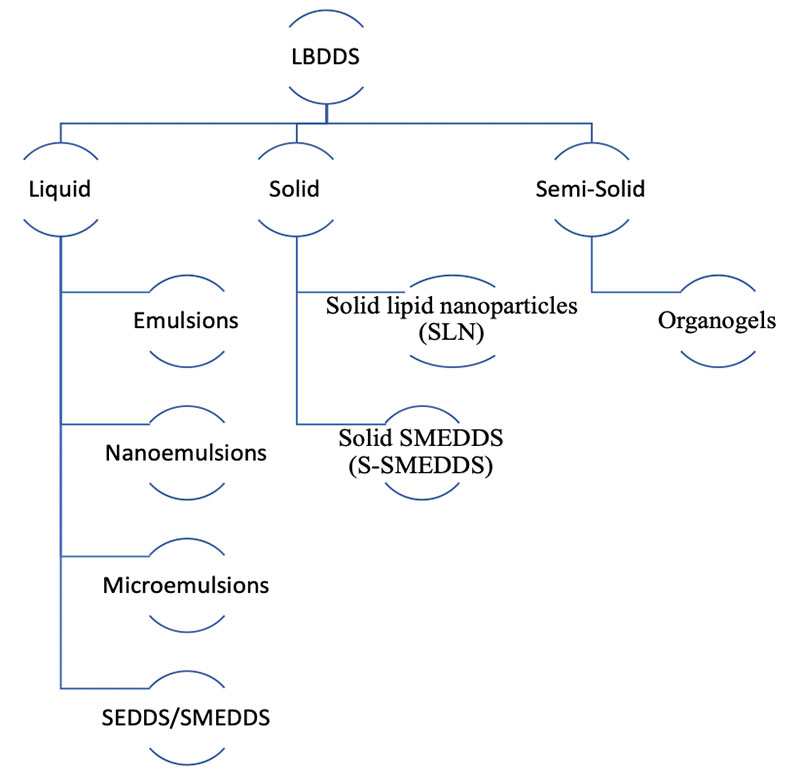
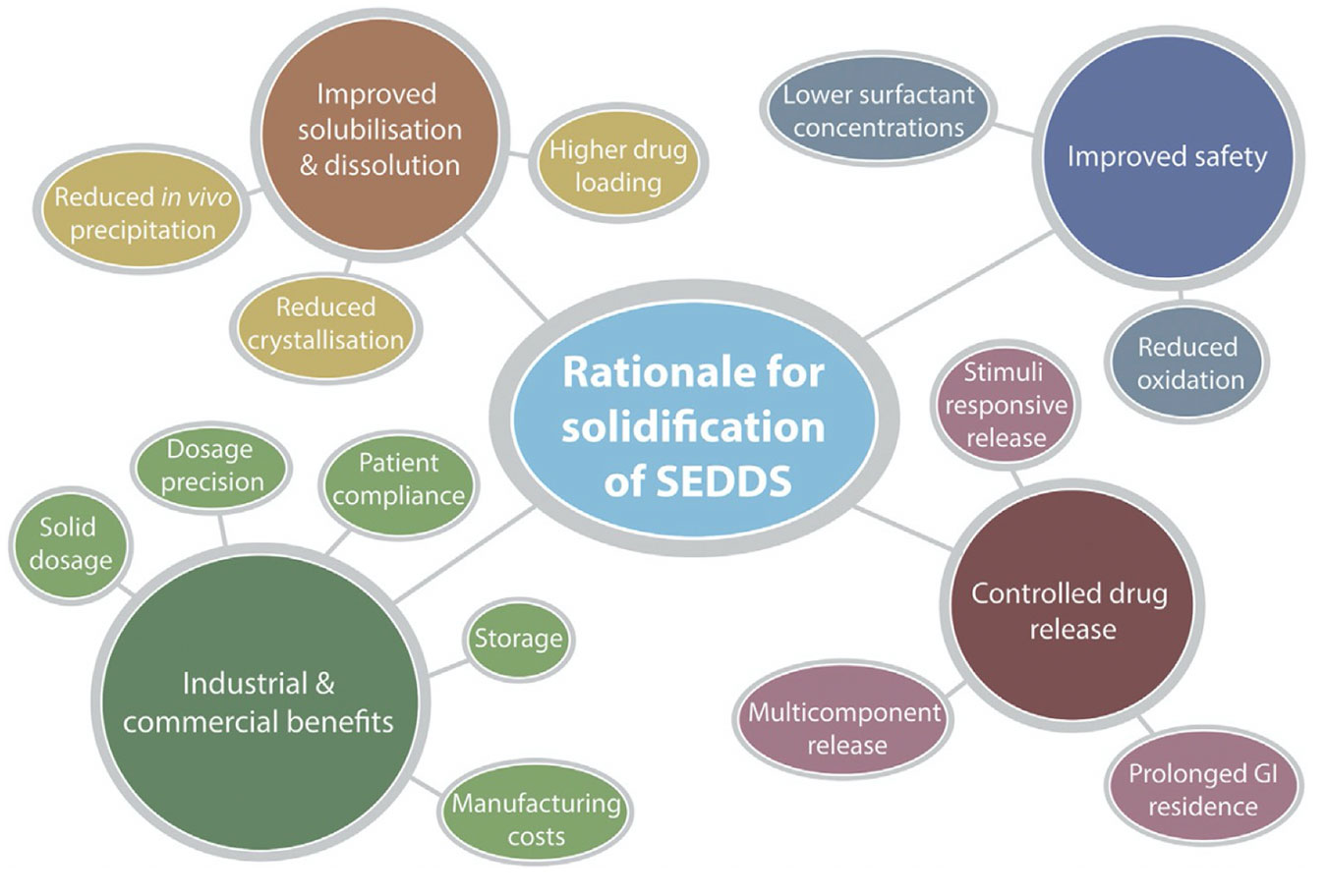
Solidification of SEDDSs affords a multitude of benefits compared to precursor liquid-SEDDSs, which can be summarised within the following categorical advantages:
- Improved drug solubilisation and dissolution,
- Improved safety,
- Controlled drug release, and
- Industrial and commercial benefits (1)

First, it allows our clients to make formulated tablet or loose powder dosage forms, the favourite mode of administration by doctors and patients. Pre-encapsulation of the whole dose into capsules/tablets provides more accurate and precise dosing in comparison to administering a prescribed volume of a suspension using a syringe or spoon and allows us to create different release profiles (immediate, delayed, controlled). It also results in better patient compliance (i.e., it is generally preferred by adult patients to administer capsules/tablets rather than a liquid formulation). The ease of use and tunability of the platform brings unique advantages to this platform in addition to the advantages of our conventional liquid SMEDDS.
Furthermore, the solidification of SMEDDSs enables simpler handling and manufacturing processes using more economic and conventional equipment (better suited to large-scale production). Solidification imparts physicochemical stability and reduces production costs while retaining, or optimising, the pharmacokinetic benefits of liquid SMEDDSs. Additionally, it avoids drug-packaging interactions, potential leakage, and migration of the excipients into the capsule shell.
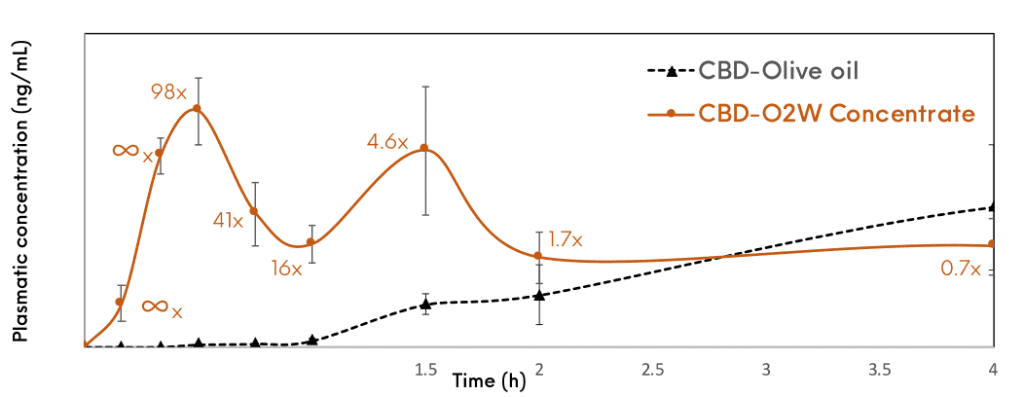
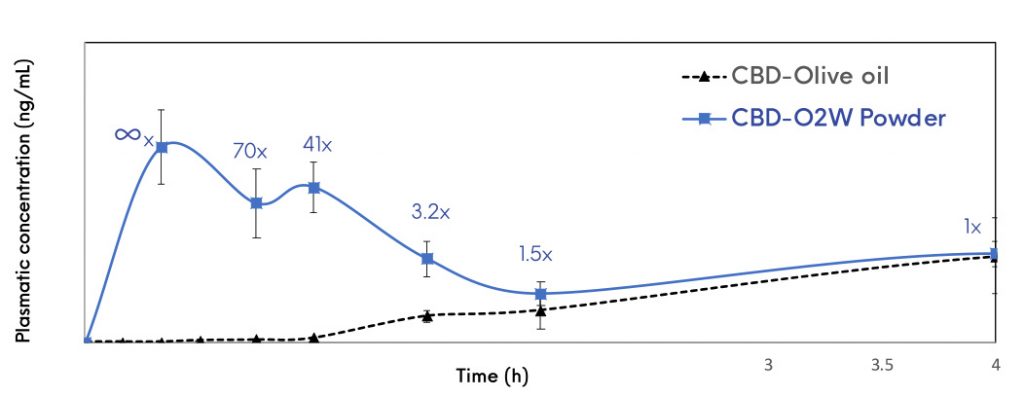
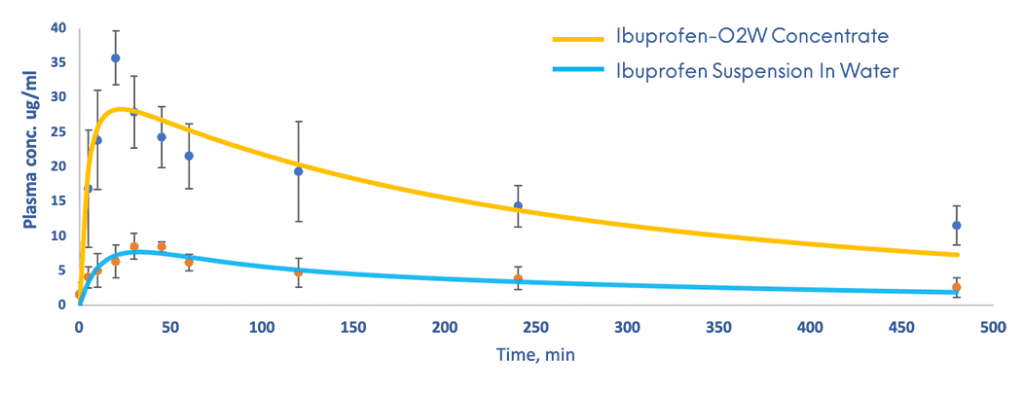
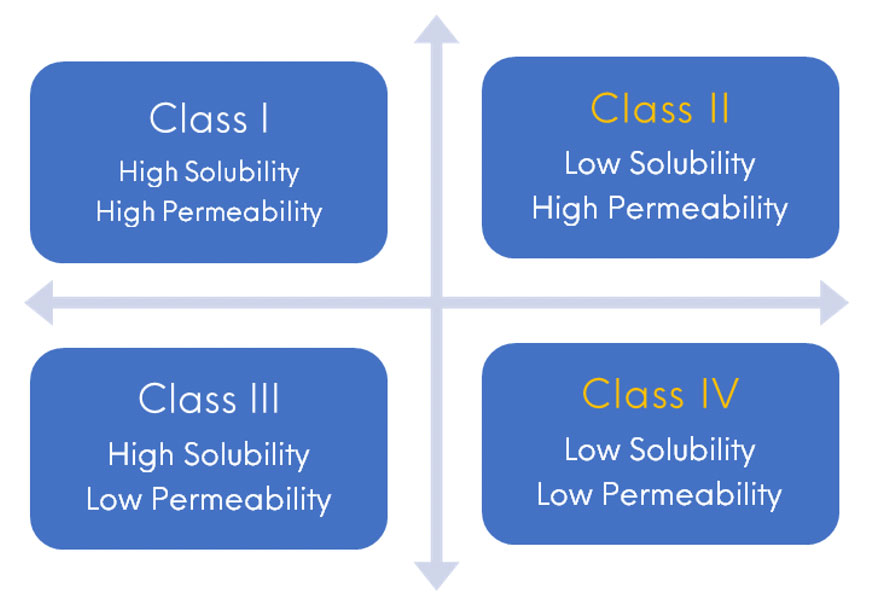
This approach has given successful results for the enhancement of solubility and bioavailability of a variety of BCS class II drugs with results significantly superior to those obtained with the pure drug during in vitro and in vivo studies. It enhances intestinal solubilisation and improves drug permeability.
It is useful to protect compounds from environmental factors like humidity, light, and oxygen. It is also useful to protect compounds that are orally administered and need to pass the gastrointestinal tract without being affected by the stomach pH and the enzymatic action. The resulting microcapsules are more stable products and easy to handle and transport, with extended shelf-life.
- Joyce, P. et al. Solidification to improve the biopharmaceutical performance of SEDDS: Opportunities and challenges. Adv. Drug Deliv. Rev. 142, 102–117 (2019).